Abstract
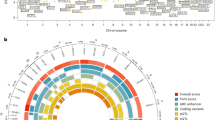
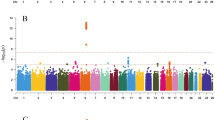
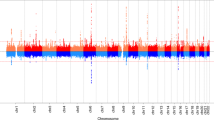
Age at menarche is a marker of timing of puberty in females. It varies widely between individuals, is a heritable trait and is associated with risks for obesity, type 2 diabetes, cardiovascular disease, breast cancer and all-cause mortality1. Studies of rare human disorders of puberty and animal models point to a complex hypothalamic-pituitary-hormonal regulation2,3, but the mechanisms that determine pubertal timing and underlie its links to disease risk remain unclear. Here, using genome-wide and custom-genotyping arrays in up to 182,416 women of European descent from 57 studies, we found robust evidence (P < 5 × 10−8) for 123 signals at 106 genomic loci associated with age at menarche. Many loci were associated with other pubertal traits in both sexes, and there was substantial overlap with genes implicated in body mass index and various diseases, including rare disorders of puberty. Menarche signals were enriched in imprinted regions, with three loci (DLK1-WDR25, MKRN3-MAGEL2 and KCNK9) demonstrating parent-of-origin-specific associations concordant with known parental expression patterns. Pathway analyses implicated nuclear hormone receptors, particularly retinoic acid and γ-aminobutyric acid-B2 receptor signalling, among novel mechanisms that regulate pubertal timing in humans. Our findings suggest a genetic architecture involving at least hundreds of common variants in the coordinated timing of the pubertal transition.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Prentice, P. & Viner, R. M. Pubertal timing and adult obesity and cardiometabolic risk in women and men: a systematic review and meta-analysis. Int. J. Obes. 37, 1036–1043 (2013)
Silveira, L. F. G. & Latronico, A. C. Approach to the patient with hypogonadotropic hypogonadism. J. Clin. Endocrinol. Metab. 98, 1781–1788 (2013)
Abreu, A. P. et al. Central precocious puberty caused by mutations in the imprinted gene MKRN3. N. Engl. J. Med. 368, 2467–2475 (2013)
Elks, C. E. et al. Thirty new loci for age at menarche identified by a meta-analysis of genome-wide association studies. Nature Genet. 42, 1077–1085 (2010)
Yang, J. et al. Conditional and joint multiple-SNP analysis of GWAS summary statistics identifies additional variants influencing complex traits. Nature Genet. 44, 369–375 (2012)
Kong, A. et al. Parental origin of sequence variants associated with complex diseases. Nature 462, 868–874 (2009)
Hindorff, L. A. et al. A catalog of published genome-wide association studies. Available at http://www.genome.gov/gwastudies. (Accessed, 1 November 2013)
Temple, I. K., Shrubb, V., Lever, M., Bullman, H. & Mackay, D. J. G. Isolated imprinting mutation of the DLK1/GTL2 locus associated with a clinical presentation of maternal uniparental disomy of chromosome 14. J. Med. Genet. 44, 637–640 (2007)
Grundberg, E. et al. Global analysis of DNA methylation variation in adipose tissue from twins reveals links to disease-associated variants in distal regulatory elements. Am. J. Hum. Genet. 93, 876–890 (2013), corrected. 93, 1158 (2013)
Westra, H.-J. et al. Systematic identification of trans eQTLs as putative drivers of known disease associations. Nature Genet. 45, 1238–1243 (2013)
Schaaf, C. P. et al. Truncating mutations of MAGEL2 cause Prader-Willi phenotypes and autism. Nature Genet. 45, 1405–1408 (2013)
Ruf, N. et al. Sequence-based bioinformatic prediction and QUASEP identify genomic imprinting of the KCNK9 potassium channel gene in mouse and human. Hum. Mol. Genet. 16, 2591–2599 (2007)
Stelzer, Y., Sagi, I., Yanuka, O., Eiges, R. & Benvenisty, N. The noncoding RNA IPW regulates the imprinted DLK1-DIO3 locus in an induced pluripotent stem cell model of Prader-Willi syndrome. Nature Genet. 46, 551–557 (2014)
Lomniczi, A. et al. Epigenetic control of female puberty. Nature Neurosci. 16, 281–289 (2013)
Partsch, C.-J. et al. Central precocious puberty in girls with Williams syndrome. J. Pediatr. 141, 441–444 (2002)
Grinspon, R. P. et al. Early onset of primary hypogonadism revealed by serum anti-Müllerian hormone determination during infancy and childhood in trisomy 21. Int. J. Androl. 34, e487–e498 (2011)
Cho, S. et al. 9-cis-Retinoic acid represses transcription of the gonadotropin-releasing hormone (GnRH) gene via proximal promoter region that is distinct from all-trans-retinoic acid response element. Brain Res. Mol. Brain Res. 87, 214–222 (2001)
Nagl, F. et al. Retinoic acid-induced nNOS expression depends on a novel PI3K/Akt/DAX1 pathway in human TGW-nu-I neuroblastoma cells. Am. J. Physiol. Cell Physiol. 297, C1146–C1156 (2009)
Zadik, Z., Sinai, T., Zung, A. & Reifen, R. Vitamin A and iron supplementation is as efficient as hormonal therapy in constitutionally delayed children. Clin. Endocrinol. 60, 682–687 (2004)
Constantin, S. et al. GnRH neuron firing and response to GABA in vitro depend on acute brain slice thickness and orientation. Endocrinology 153, 3758–3769 (2012)
Speliotes, E. K. et al. Association analyses of 249,796 individuals reveal 18 new loci associated with body mass index. Nature Genet. 42, 937–948 (2010)
Lango Allen, H. et al. Hundreds of variants clustered in genomic loci and biological pathways affect human height. Nature 467, 832–838 (2010)
Horikoshi, M. et al. New loci associated with birth weight identify genetic links between intrauterine growth and adult height and metabolism. Nature Genet. 45, 76–82 (2013)
D’Aloisio, A. A., DeRoo, L. A., Baird, D. D., Weinberg, C. R. & Sandler, D. P. Prenatal and infant exposures and age at menarche. Epidemiology 24, 277–284 (2013)
Barrett, J. C. et al. Genome-wide association study and meta-analysis find that over 40 loci affect risk of type 1 diabetes. Nature Genet. 41, 703–707 (2009)
Zheng, W. et al. Genome-wide association study identifies a new breast cancer susceptibility locus at 6q25.1. Nature Genet. 41, 324–328 (2009)
Estrada, K. et al. Genome-wide meta-analysis identifies 56 bone mineral density loci and reveals 14 loci associated with risk of fracture. Nature Genet. 44, 491–501 (2012)
Parker, S. E. et al. Menarche, menopause, years of menstruation, and the incidence of osteoporosis: the influence of prenatal exposure to diethylstilbestrol. J. Clin. Endocrinol. Metab. 99, 594–601 (2014)
Huang, J. et al. Genome-wide association study for circulating levels of PAI-1 provides novel insights into its regulation. Blood 120, 4873–4881 (2012)
Migliano, A. B., Vinicius, L. & Lahr, M. M. Life history trade-offs explain the evolution of human pygmies. Proc. Natl Acad. Sci. USA 104, 20216–20219 (2007)
Michailidou, K. et al. Large-scale genotyping identifies 41 new loci associated with breast cancer risk. Nature Genet. 45, 353–361 (2013)
Willer, C. J., Li, Y. & Abecasis, G. R. METAL: fast and efficient meta-analysis of genomewide association scans. Bioinformatics 26, 2190–2191 (2010)
Yang, J. et al. Genomic inflation factors under polygenic inheritance. Eur. J. Hum. Genet. 19, 807–812 (2011)
Yang, J., Lee, S. H., Goddard, M. E. & Visscher, P. M. GCTA: a tool for genome-wide complex trait analysis. Am. J. Hum. Genet. 88, 76–82 (2011)
The InterAct Consortium Design and cohort description of the InterAct Project: an examination of the interaction of genetic and lifestyle factors on the incidence of type 2 diabetes in the EPIC Study. Diabetologia 54, 2272–2282 (2011)
Göring, H. H. H. et al. Discovery of expression QTLs using large-scale transcriptional profiling in human lymphocytes. Nature Genet. 39, 1208–1216 (2007)
Idaghdour, Y. et al. Geographical genomics of human leukocyte gene expression variation in southern Morocco. Nature Genet. 42, 62–67 (2010)
Heap, G. A. et al. Complex nature of SNP genotype effects on gene expression in primary human leucocytes. BMC Med. Genomics 2, 1 (2009)
Emilsson, V. et al. Genetics of gene expression and its effect on disease. Nature 452, 423–428 (2008)
Fehrmann, R. S. N. et al. Trans-eQTLs reveal that independent genetic variants associated with a complex phenotype converge on intermediate genes, with a major role for the HLA. PLoS Genet. 7, e1002197 (2011)
Mehta, D. et al. Impact of common regulatory single-nucleotide variants on gene expression profiles in whole blood. Eur. J. Hum. Genet. 21, 48–54 (2013)
Maeda, T. et al. The correlation between clinical laboratory data and telomeric status of male patients with metabolic disorders and no clinical history of vascular events. Aging Male 14, 21–26 (2011)
Sasayama, D. et al. Identification of single nucleotide polymorphisms regulating peripheral blood mRNA expression with genome-wide significance: an eQTL study in the Japanese population. PLoS ONE 8, e54967 (2013)
Dixon, A. L. et al. A genome-wide association study of global gene expression. Nature Genet. 39, 1202–1207 (2007)
Liang, L. et al. A cross-platform analysis of 14,177 expression quantitative trait loci derived from lymphoblastoid cell lines. Genome Res. 23, 716–726 (2013)
Stranger, B. E. et al. Population genomics of human gene expression. Nature Genet. 39, 1217–1224 (2007)
Kwan, T. et al. Genome-wide analysis of transcript isoform variation in humans. Nature Genet. 40, 225–231 (2008)
Dimas, A. S. et al. Common regulatory variation impacts gene expression in a cell type-dependent manner. Science 325, 1246–1250 (2009)
Cusanovich, D. A. et al. The combination of a genome-wide association study of lymphocyte count and analysis of gene expression data reveals novel asthma candidate genes. Hum. Mol. Genet. 21, 2111–2123 (2012)
Grundberg, E. et al. Mapping cis- and trans-regulatory effects across multiple tissues in twins. Nature Genet. 44, 1084–1089 (2012)
Fairfax, B. P. et al. Genetics of gene expression in primary immune cells identifies cell type-specific master regulators and roles of HLA alleles. Nature Genet. 44, 502–510 (2012)
Murphy, A. et al. Mapping of numerous disease-associated expression polymorphisms in primary peripheral blood CD4+ lymphocytes. Hum. Mol. Genet. 19, 4745–4757 (2010)
Heinzen, E. L. et al. Tissue-specific genetic control of splicing: implications for the study of complex traits. PLoS Biol. 6, e1 (2008)
Zeller, T. et al. Genetics and beyond–the transcriptome of human monocytes and disease susceptibility. PLoS ONE 5, e10693 (2010)
Barreiro, L. B. et al. Deciphering the genetic architecture of variation in the immune response to Mycobacterium tuberculosis infection. Proc. Natl Acad. Sci. USA 109, 1204–1209 (2012)
Huang, R. S. et al. Population differences in microRNA expression and biological implications. RNA Biol. 8, 692–701 (2011)
Degner, J. F. et al. DNase I sensitivity QTLs are a major determinant of human expression variation. Nature 482, 390–394 (2012)
Greenawalt, D. M. et al. A survey of the genetics of stomach, liver, and adipose gene expression from a morbidly obese cohort. Genome Res. 21, 1008–1016 (2011)
Kompass, K. S. & Witte, J. S. Co-regulatory expression quantitative trait loci mapping: method and application to endometrial cancer. BMC Med. Genomics 4, 6 (2011)
Li, Q. et al. Integrative eQTL-based analyses reveal the biology of breast cancer risk loci. Cell 152, 633–641 (2013)
Webster, J. A. et al. Genetic control of human brain transcript expression in Alzheimer disease. Am. J. Hum. Genet. 84, 445–458 (2009)
Zou, F. et al. Brain expression genome-wide association study (eGWAS) identifies human disease-associated variants. PLoS Genet. 8, e1002707 (2012)
Colantuoni, C. et al. Temporal dynamics and genetic control of transcription in the human prefrontal cortex. Nature 478, 519–523 (2011)
Liu, C. et al. Whole-genome association mapping of gene expression in the human prefrontal cortex. Mol. Psychiatry 15, 779–784 (2010)
Gibbs, J. R. et al. Abundant quantitative trait loci exist for DNA methylation and gene expression in human brain. PLoS Genet. 6, e1000952 (2010)
Zhang, B. et al. Integrated systems approach identifies genetic nodes and networks in late-onset Alzheimer’s disease. Cell 153, 707–720 (2013)
Schadt, E. E. et al. Mapping the genetic architecture of gene expression in human liver. PLoS Biol. 6, e107 (2008)
Innocenti, F. et al. Identification, replication, and functional fine-mapping of expression quantitative trait loci in primary human liver tissue. PLoS Genet. 7, e1002078 (2011)
Sulzbacher, S., Schroeder, I. S., Truong, T. T. & Wobus, A. M. Activin A-induced differentiation of embryonic stem cells into endoderm and pancreatic progenitors-the influence of differentiation factors and culture conditions. Stem Cell Rev. 5, 159–173 (2009)
Schröder, A. et al. Genomics of ADME gene expression: mapping expression quantitative trait loci relevant for absorption, distribution, metabolism and excretion of drugs in human liver. Pharmacogenomics J. 13, 12–20 (2013)
Grundberg, E. et al. Population genomics in a disease targeted primary cell model. Genome Res. 19, 1942–1952 (2009)
Kabakchiev, B. & Silverberg, M. S. Expression quantitative trait loci analysis identifies associations between genotype and gene expression in human intestine. Gastroenterology 144, 1488–1496e3 (2013)
Hao, K. et al. Lung eQTLs to help reveal the molecular underpinnings of asthma. PLoS Genet. 8, e1003029 (2012)
Ding, J. et al. Gene expression in skin and lymphoblastoid cells: refined statistical method reveals extensive overlap in cis-eQTL signals. Am. J. Hum. Genet. 87, 779–789 (2010)
Rantalainen, M. et al. MicroRNA expression in abdominal and gluteal adipose tissue is associated with mRNA expression levels and partly genetically driven. PLoS ONE 6, e27338 (2011)
Segrè, A. V., Groop, L., Mootha, V. K., Daly, M. J. & Altshuler, D. Common inherited variation in mitochondrial genes is not enriched for associations with type 2 diabetes or related glycemic traits. PLoS Genet. 6, e1001058 (2010)
Cousminer, D. L. et al. Genome-wide association study of sexual maturation in males and females highlights a role for body mass and menarche loci in male puberty. Hum. Mol. Genet.; Epub ahead of print. (2014)
Acknowledgements
A full list of acknowledgements can be found in the Supplementary Information.
Author information
Authors and Affiliations
Consortia
Contributions
Overall project management: J.R.B.P., F.D., C.E.E., P.S., D.J.T., D.F.E., K.S., J.M.M. and K.K.O. Core analyses: J.R.B.P., F.D., C.E.E., P.S., T.F., D.J.T., D.I.C. and T.E. Individual study analysts: A.A.R., A.D., A.G., A.J., A.T., A.V.S., B.Z.A., B.F., C.E.E., D.F.G., D.I.C., D.J.T., D.L.C., D.L.K., E.A., E.K.W., E.M., E.M.B., E.T., F.D., G.M., G.McMahon, I.M.N., J.A.V., J.D., J.H., J.R.B.P., J.T., J.Z., K.L.L., K.M., L.L.P., L.M.R., L.M.Y., L.S., M.M., N.F., N.Ts., P.K., P.S., R.M., S.K., S.S., S.S.U., T.C., T.E., T.F., T.Fo., T.H.P., W.Q.A. and Z.K. Individual study data management and generation: A.A.R., A.C.H., A.D., A.D.C., A.G.U., A.J.O., A.M.S., A.Mu., A.P., A.Po., B.A.O., C.A.H., D.C., D.I.C., D.J.H., D.K., D.Lw., D.P.K., D.P.S., D.S., E.A.N., E.P., E.W., F.A., F.B.H., F.G., F.R., G.D., G.E., G.G.W., H.S., H.W., I.D., J.C., J.H., J.P.R., L.F., L.Fr., L.M., L.M.R., M.E.G., M.J.S., M.J.W., M.K.B., M.Melbye, M.P., M.W., N.A., N.J.T., N.L.P., P.K.M., Q.W., R.H., S.B., S.C., S.G., S.L., S.R., S.S.U., T.E., U.S., U.T., V.S. and W.L.M. Individual study principal investigators: A.C., A.G.U., A.H., A.J.O., A.K.D., A.L., A.M., A.M.D., A.Mannermaa, A.Mu., A.R., B.B., B.Z.A., B.H.R.W., C.B., C.E.P., C.G., C.H., C.van Duijn, D.I.B., D.F., D.F.E., D.J.H., D.L., D.Lw., D.S.P., D.P.S., D.Schlessinger, E.A.S., E.B., E.E.J.d.G., E.I., E.W., E.W.D., F.B.H., F.J.C., G.C., G.D., G.G.G., G.Wa., G.Wi., G.W.M., H.A., H.A.B., H.B., H.Be., H.F., H.N., H.S., H.V., I.D., I.L.A., J.A.K., J.B., J.C.C., J.G.E., J.E.B., J.L.H., J.M.C., J.M.M., J.P., K.C., K.K., K.K.O., K.P., K.S., L.C., L.F., L.J.B., M.C.S., M.G., M.I.M., M.J., M.J.E., M.J.H., M.J.S., M.K.S., M.W.B., M.Z., N.G.M., N.J.W., P.A.F., P.D., P.D.P.P., P.F.M., P.G., P.H., P.K., P.M.R., P.N., P.P., P.P.G., P.R., P.V., R.J.F.L., R.L.M., R.W., S.B., S.Bergmann, S.C., S.E.B., T.B.H., T.D.S., T.I.A.S., U.H., V.G., V.K. and V.S.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Plots of all 106 menarche loci and genome-wide summary level statistics are available at the ReproGen Consortium website: http://www.reprogen.org.
Extended data figures and tables
Extended Data Figure 2 Estimates of genetic variance explained.
Variance in age at menarche in the EPIC-InterAct replication sample (N = 8,689) explained by combined sets of SNPs defined by their strength of association in the discovery set.
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-5 and 8 and 9. (PDF 1373 kb)
Supplementary Data
This file contains Supplementary Tables 6 and 7. (XLSX 323 kb)
Rights and permissions
About this article
Cite this article
Perry, J., Day, F., Elks, C. et al. Parent-of-origin-specific allelic associations among 106 genomic loci for age at menarche. Nature 514, 92–97 (2014). https://doi.org/10.1038/nature13545
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature13545
This article is cited by
-
Identifying causal associations between women’s reproductive traits and risk of schizophrenia: a multivariate validated two-sample Mendelian randomization analysis
BMC Psychiatry (2024)
-
Mendelian randomization identifies circulating proteins as biomarkers for age at menarche and age at natural menopause
Communications Biology (2024)
-
The impact of reproductive factors on the metabolic profile of females from menarche to menopause
Nature Communications (2024)
-
Cloning, tissue expression and imprinting status analysis of the NDN gene in Dolang sheep
Molecular Biology Reports (2024)
-
Genetic Prediction of Osteoporosis by Anti-Müllerian Hormone Levels and Reproductive Factors in Women: A Mendelian Randomization Study
Calcified Tissue International (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.