Abstract
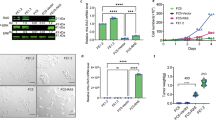
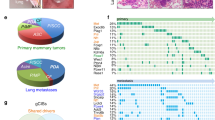
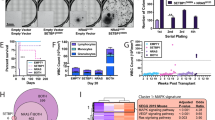
B-lymphoma Moloney murine leukaemia virus insertion region-1 (BMI1) is a member of the polycomb group of transcription repressors, which functions in stem cell maintenance and oncogenesis through the inhibition of the INK4A/ARF tumour suppressor locus. Overexpression of BMI1 is associated with poor prognosis in several human cancers, including breast cancer. We have previously shown that BMI1 collaborates with H-RAS to induce transformation of MCF10A human mammary epithelial cells through dysregulation of multiple growth pathways independent of the INK4A/ARF locus. In this study, we show that BMI1 collaborates with H-RAS to promote increased proliferation, invasion and resistance to apoptosis in vitro, and an increased rate of spontaneous metastases from mammary fat pad xenografts including novel metastases to the brain. Furthermore, in collaboration with H-RAS, BMI1 induced fulminant metastatic disease in the lung using a tail vein model of haematogenous spread through accelerated cellular proliferation and inhibition of apoptosis. Finally, we show that knockdown of BMI1 in several established breast cancer cell lines leads to decreased oncogenic behaviour in vitro and in vivo. In summary, BMI1 collaborates with H-RAS to induce an aggressive and metastatic phenotype with the unusual occurrence of brain metastasis, making it an important target for diagnosis and treatment of aggressive breast cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
251,40 € per year
only 5,03 € per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Bendell JC, Domchek SM, Burstein HJ, Harris L, Younger J, Kuter I et al. (2003). Central nervous system metastases in women who receive trastuzumab-based therapy for metastatic breast carcinoma. Cancer 97: 2972–2977.
Brackstone M, Townson JL, Chambers AF . (2007). Tumour dormancy in breast cancer: an update. Breast Cancer Res 9: 208.
Campbell MJ, Esserman LJ, Zhou Y, Shoemaker M, Lobo M, Borman E et al. (2006). Breast cancer growth prevention by statins. Cancer Res 66: 8707–8714.
Clayton AJ, Danson S, Jolly S, Ryder WD, Burt PA, Stewart AL et al. (2004). Incidence of cerebral metastases in patients treated with trastuzumab for metastatic breast cancer. Br J Cancer 91: 639–643.
Datta S, Hoenerhoff MJ, Bommi P, Sainger R, Guo W-J, Dimri M et al. (2007). Bmi-1 Cooperates with H-Ras to Transform Human Mammary Epithelial Cells via Dysregulation of Multiple Growth-Regulatory Pathways. Cancer Res 67: 10286–10295.
Debnath J, Mills KR, Collins NL, Reginato MJ, Muthuswamy SK, Brugge JS . (2002). The role of apoptosis in creating and maintaining luminal space within normal and oncogene-expressing mammary acini. Cell 111: 29–40.
Desprez PY, Lin CQ, Thomasset N, Sympson CJ, Bissell MJ, Campisi J . (1998). A novel pathway for mammary epithelial cell invasion induced by the helix-loop-helix protein Id-1. Mol Cell Biol 18: 4577–4588.
Dimri GP, Martinez JL, Jacobs JJ, Keblusek P, Itahana K, Van Lohuizen M et al. (2002). The Bmi-1 oncogene induces telomerase activity and immortalizes human mammary epithelial cells. Cancer Res 62: 4736–4745.
Douglas D, Hsu JH, Hung L, Cooper A, Abdueva D, van Doorninck J et al. (2008). BMI-1 promotes ewing sarcoma tumorigenicity independent of CDKN2A repression. Cancer Res 68: 6507–6515.
Feng Y, Song LB, Guo BH, Liao WT, Li MZ, Liu WL et al. (2007). [Expression and significance of Bmi-1 in breast cancer]. Ai Zheng 26: 154–157.
Fisher B, Jeong JH, Dignam J, Anderson S, Mamounas E, Wickerham DL et al. (2001). Findings from recent National Surgical Adjuvant Breast and Bowel Project adjuvant studies in stage I breast cancer. J Natl Cancer Inst Monogr 30: 62–66.
Glinsky GV . (2005). Death-from-cancer signatures and stem cell contribution to metastatic cancer. Cell Cycle 4: 1171–1175.
Glinsky GV . (2006). Genomic models of metastatic cancer: functional analysis of death-from-cancer signature genes reveals aneuploid, anoikis-resistant, metastasis-enabling phenotype with altered cell cycle control and activated Polycomb Group (PcG) protein chromatin silencing pathway. Cell Cycle 5: 1208–1216.
Glinsky GV, Berezovska O, Glinskii AB . (2005). Microarray analysis identifies a death-from-cancer signature predicting therapy failure in patients with multiple types of cancer. J Clin Invest 115: 1503–1521.
Haupt Y, Alexander WS, Barri G, Klinken SP, Adams JM . (1991). Novel zinc finger gene implicated as myc collaborator by retrovirally accelerated lymphomagenesis in E mu-myc transgenic mice. Cell 65: 753–763.
Haupt Y, Bath ML, Harris AW, Adams JM . (1993). bmi-1 transgene induces lymphomas and collaborates with myc in tumorigenesis. Oncogene 8: 3161–3164.
He B, Mirza M, Weber GF . (2006). An osteopontin splice variant induces anchorage independence in human breast cancer cells. Oncogene 25: 2192–2202.
Huang KH, Liu JH, Li XX, Song LB, Zeng MS . (2007). Association of Bmi-1 mRNA expression with differentiation, metastasis and prognosis of gastric carcinoma. Nan Fang Yi Ke Da Xue Xue Bao 27: 973–975.
Jacobs JJ, Scheijen B, Voncken JW, Kieboom K, Berns A, van Lohuizen M . (1999). Bmi-1 collaborates with c-Myc in tumorigenesis by inhibiting c-Myc-induced apoptosis via INK4a/ARF. Genes Dev 13: 2678–2690.
Kang MK, Kim RH, Kim SJ, Yip FK, Shin KH, Dimri GP et al. (2007). Elevated Bmi-1 expression is associated with dysplastic cell transformation during oral carcinogenesis and is required for cancer cell replication and survival. Br J Cancer 96: 126–133.
Kim JH, Yoon SY, Jeong SH, Kim SY, Moon SK, Joo JH et al. (2004). Overexpression of Bmi-1 oncoprotein correlates with axillary lymph node metastases in invasive ductal breast cancer. Breast 13: 383–388.
Lessard J, Sauvageau G . (2003). Bmi-1 determines the proliferative capacity of normal and leukaemic stem cells. Nature 423: 255–260.
Liu S, Dontu G, Mantle ID, Patel S, Ahn NS, Jackson KW et al. (2006). Hedgehog signaling and Bmi-1 regulate self-renewal of normal and malignant human mammary stem cells. Cancer Res 66: 6063–6071.
Mihic-Probst D, Kuster A, Kilgus S, Bode-Lesniewska B, Ingold-Heppner B, Leung C et al. (2007). Consistent expression of the stem cell renewal factor BMI-1 in primary and metastatic melanoma. Int J Cancer 121: 1764–1770.
Mukhopadhyay R, Theriault RL, Price JE . (1999). Increased levels of alpha6 integrins are associated with the metastatic phenotype of human breast cancer cells. Clin Exp Metastasis 17: 325–332.
Price JE . (1996). Metastasis from human breast cancer cell lines. Breast Cancer Res Treat 39: 93–102.
Song LB, Zeng MS, Liao WT, Zhang L, Mo HY, Liu WL et al. (2006). Bmi-1 is a novel molecular marker of nasopharyngeal carcinoma progression and immortalizes primary human nasopharyngeal epithelial cells. Cancer Res 66: 6225–6232.
Tsukada Y, Fouad A, Pickren JW, Lane WW . (1983). Central nervous system metastasis from breast carcinoma. Autopsy study. Cancer 52: 2349–2354.
van Kemenade FJ, Raaphorst FM, Blokzijl T, Fieret E, Hamer KM, Satijn DP et al. (2001). Coexpression of BMI-1 and EZH2 polycomb-group proteins is associated with cycling cells and degree of malignancy in B-cell non-Hodgkin lymphoma. Blood 97: 3896–3901.
Vernon AE, Bakewell SJ, Chodosh LA . (2007). Deciphering the molecular basis of breast cancer metastasis with mouse models. Rev Endocr Metab Disord 8: 199–213.
Wallgren A, Bonetti M, Gelber RD, Goldhirsch A, Castiglione-Gertsch M, Holmberg SB et al. (2003). Risk factors for locoregional recurrence among breast cancer patients: results from International Breast Cancer Study Group Trials I through VII. J Clin Oncol 21: 1205–1213.
Acknowledgements
We are grateful to Max Wicha for providing the shRNA lentiviral constructs, George Dunn, Asa Dorsey and Gail McMullen for animal care, Zi-ayo Liu for lentiviral transductions and Suresh Arya for advice on lentiviral preparation, and Aleksandra Michalowski and the NIH Biostatisitics Branch for assistance with statistical analysis. This work was supported in part by the Intramural Program of the NIH, Center for Cancer Research, National Cancer Institute and by RO1CA 094150 (GD).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Rights and permissions
About this article
Cite this article
Hoenerhoff, M., Chu, I., Barkan, D. et al. BMI1 cooperates with H-RAS to induce an aggressive breast cancer phenotype with brain metastases. Oncogene 28, 3022–3032 (2009). https://doi.org/10.1038/onc.2009.165
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.165